In England, copper-silver ionization is applied in about 120 hospitals successfully for the deactivation of Legionella bacteria. The ion generator that was used, was the size of a matchbox.īecause of copper-silver ionization, drinking water could be produced safely in space without the use of chlorine.įigure 2: NASA used Apollo flights for one of the first copper-silver ionizations 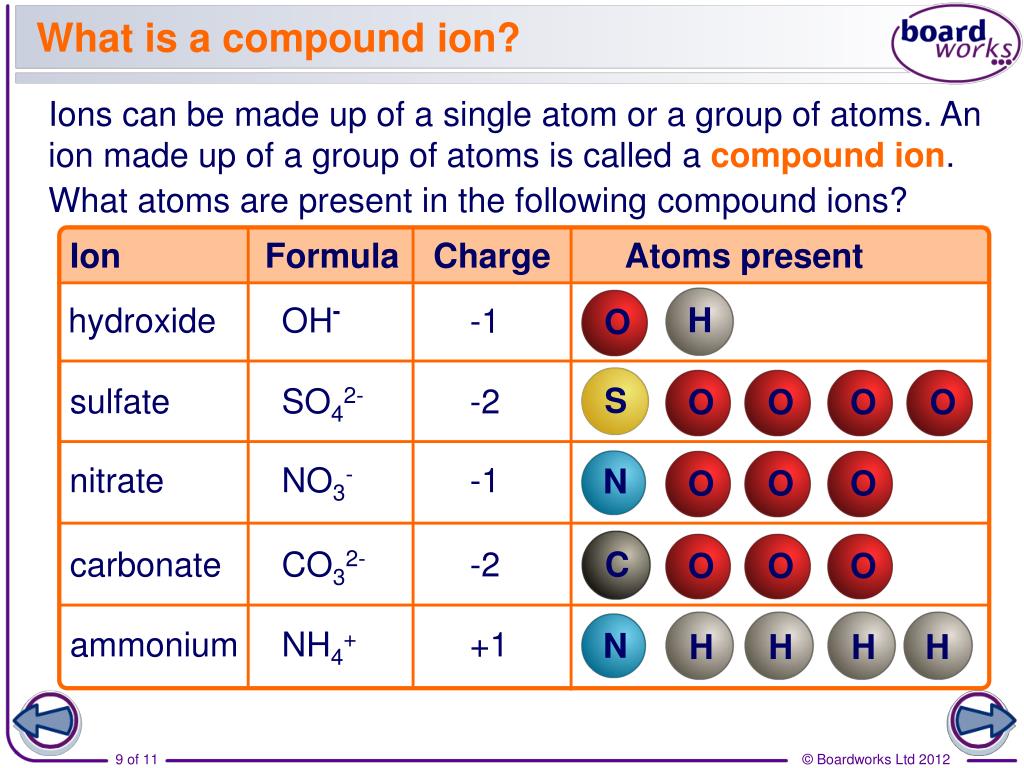
It became of interest when NASA used copper-silver ionization for drinking water prodcution aboard Apollo space ships in 1960. What are the applications of copper-silver ionization?Ĭopper-silver ionization is suitable for a large number of applications. Copper (Cu +) ions are unstable in water, unless a stabilizing ligand is present. Copper can be found in the water in free form. When copper ions (Cu +) dissolve in water, they are oxidized immediately to form Cu 2+ ions. The number of ions that is released increases, when electric charges are higher. The ion concentration is determined by the water flow. Generally the amount of silver ions at a copper ion rate of 0,15 to 0,40 ppm lies between 5 and 50 ppb. 
The larger part of the ions flows away through the water, before reaching the opposite electrode. An electric current is created, causing the outer atoms of the electrodes to lose an electron and become positively charged. The water that is disinfected flows past the electrodes. The electrodes are placed close together. When copper-silver ionization is applied, positively charged copper (Cu + and Cu 2+) and silver (Ag +) ions are formed. During ionization, atoms turn into kations or anions. A positively charged ion in called a kation and a negatively charged ion is calles an anion. An electric current is created through copper-silver, causing positively charged copper and silver ions to form.Ĭopper-silver ionization brings us back to basic chemistry: an ion an electrically charged atom, has a positive charge when it gives up an electron and a negative charge when it takes up an electron. Some European and Russian villages have been using silver for drinking water treatment for many years.Ĭopper-silver ionization was developed in both Europe and the United States in the 1950’s.įigure 1: the Vickings used copper strings to prevent algal growth on shipsĬopper-silver ionization is brought about by electrolysis. Since 1869 various publications have appeared on disinfection properties of silver. Well water containing copper and silver coins is very bright, due to the biocidal affect of these metals. Nomads used silver coins to improve drinking water quality. Because of this measure, ships can reach their destination faster. Most anti-fouling paints contain copper, reducing the number of marine species growing on the walls of ships. Modern ships still use the same technology. The Vickings used copper strings on their ships to prevent the growth of algae and shells. Silver can be obtained from pure deposits, from silver ores such as argenite (Ag 2S) and horn silver (AgCl) and combined with ore deposites that contain lead, gold or copper.īoth copper and silver have been applied for centuries because of their biocidal mechanism. Large deposits of copper ores have been found throughout the US, Chili, Zambia, Zaïre, peru and Canada. Nowadays copper is mainly extracted form ores, such as cuprite (CuO 2), tenorite (CuO), malachite (CuO 3♼u(OH) 2), chalcocite (Cu 2S), covelite (CuS) and bornite (Cu 6FeS 4). The Roman empire gained most of its copper from Cyprus, the isle that gave copper its name. More dan 7000 years ago people developed a copper extraction mechanism for copper ores. Copper can be easily extracted and processed. When was the disinfection mechanism of copper and silver discovered?Īrcheological excavations show, that people have been using copper for more than 11.000 years and have been using silver for more than 5000 years. Metals such as copper and silver can be used for water disinfection, if they are ionized. Separation and Concentration Purification Request.Plant Inspection & Process Optimalisation. 
Lund (Anoka-Ramsey Community College), Melissa Alviar-Agnew, and Henry Agnew. This page is shared under a CK-12 license and was authored, remixed, and/or curated by Lance S. \): The Modern and Common System of Cation Names Element 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
